 
Examples similar to this one can be found throughout the solar system, particularly in the Gas Giants (Benner 680). Therefore it would be reasonable to consider the atmosphere of Venus a supercritical fluid because both the pressure and temperature exceed that of Carbon Dioxide's critical point however this theory has not been proven. The atmospheric pressure of Venus is approximately 90 times greater than that of the Earth, with an average temperature of 467 ☌, and about 97% of its atmosphere is Carbon Dioxide. For instance, water that is supercritical differs from regular water in the fact that it is non-polar and acidic (Benner 680). The densities and viscosity of a supercritical fluid are subject to change when pressure or temperature are tampered with, and the supercritical fluid of a substance can have very different properties than the regular fluids. Supercritical fluids have no surface tension because they are not subject to the vapor-liquid boundary so no molecules have the attraction to the interior of the liquid. The Mole Fraction Solubility is simply the ratio of the sublimation (or vapor pressure) to the total gas pressure. As can be noted from the Mole Fraction Solubility. It becomes especially easy to dissolve liquids and solids such as these in a supercritical fluid because of the high density. 
Volatile liquids and solids, or liquids and solids with a high vapor pressure or low boiling point, are soluble in gas. However, it is possible to go from a gas to a liquid without crossing the boundary between the vapor and liquid phase using a supercritical fluid just by lowering the temperature of the liquid (Observe phase diagram below). Supercritical fluids have the low viscosityof a gas and the high density of a liquid, making it impossible to liquefy the matter using any amount of pressure. In 1822 Baron Charles Cagniard de la Tour discovered supercritical fluids while conducting experiments involving the discontinuities of the sound of a flint ball in a sealed cannon barrel filled with various fluids at various temperatures ("Charles Cagniard de la Tour"). 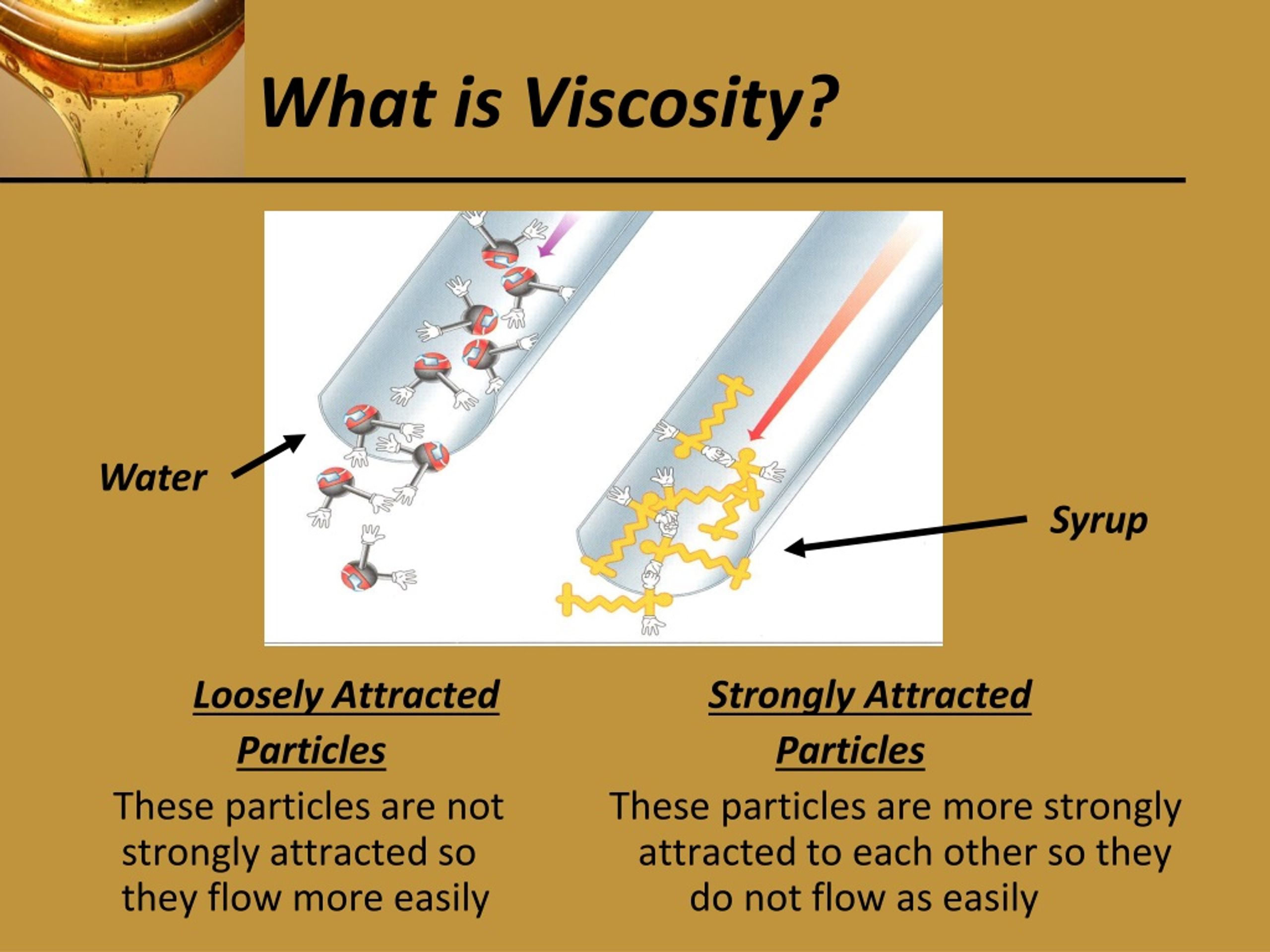
This stage is characterized by the inability to distinguish whether the matter is a liquid or a gas, as a result, Supercritical fluids (SCF) do not have a definite phase. Matter can be pushed to temperatures and pressures beyond those of its critical point. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
